September 16 2016
Third in a series of blogs reprinted by permission of the author from the website Vaccine Papers. The original is located here. This paper covers the experimental evidence demonstrating that the same prenatal immune activation that causes autistic behavior and brain changes in mice, as well as high levels of IL-6 cytokines, a necessary and sufficient condition for the development of autism, causes similarly high levels of IL-6 and autistic behaviors in primates, which are much more biologically similar to humans than mice are.
All the studies described in parts 1 and 2 use mice. Autism is always diagnosed by behavior; there is at present no objective blood test for autism. Consequently, some people question the diagnosis of “autism” in mice, since mouse behavior is so different from human behavior. This is a reasonable concern.
Responding to this reasonable concern, the Patterson group has repeated the maternal immune activation experiments with monkeys. Monkeys are an excellent animal to use in autism experiments, because they have much more complex social interactions, more like humans.
Monkeys are difficult to use in research, however. They are much more expensive than mice. They require more space and more time than mice. That’s why the mouse studies were completed first. Monkey studies were not pursued until there was a sufficient level of understanding of immune activation. This is normal research progression. It’s unwise to perform experiments with monkeys before effects in mouse models are thoroughly understood.
Three papers have been published on immune activation experiments in monkeys, all by the Patterson group at Cal Tech:
Paper 1: “Activation of the Maternal Immune System during Pregnancy Alters Behavioral Development of Rhesus Monkey Offspring”
Paper 2: “Maternal Immune Activation in Nonhuman Primates Alters Social Attention in Juvenile Offspring”
Paper 3: “Preliminary Evidence of Neuropathology in Nonhuman Primates Prenatally Exposed to Maternal Immune Activation”
The first two papers describe the behavioral tests only. The third paper reports the first, preliminary physical investigations of dissected monkey brains, which found that immune activation caused changes in neuron features (changes in dendrite density and dendrite diameter in specific locations).
Methods
Poly(I:C) was used to stimulate immune activation in pregnant rhesus monkeys. Immune activation occurred either during late first trimester (MIA1 monkeys), or late second trimester (MIA2 monkeys). A control group received saline injections. Monkeys received three injections, spread out over four days. MIA = maternal immune activation.
Pregnant monkeys injected with poly(I:C) displayed fever, cytokine production, and general sickness behavior. IL-6 levels were measured in blood. (See paper #3 for the IL-6 results. IL-6 ranged between 3,635 and 23,441 pg/mL three hours after the first dose. These are very high IL-6 concentrations.)
Offspring monkeys were then subjected to behavioral tests as infants and as juveniles.
Attachment Test
This test is performed with weaned infants, at 6-12 months of age. Infants are separated from their mothers for four days and observed for “self-soothing” behaviors (e.g. tantrums). Results are shown below.
Above: Immune activation caused changes in how infant monkeys respond to being separated from their mothers. Immune activation caused either an increase (MIA-2) or decrease (MIA-1) in vocalization during separation.
Interestingly, MIA1 and MIA2 monkeys displayed opposite behavior changes. First trimester immune activation exposure caused a large reduction in self-soothing; second trimester immune activation exposure caused a large increase in self-soothing.
The paper states
The MIA2 offspring displayed a dramatic increase in distress/self-soothing behaviors over the 4- day testing period that was not observed in the control animals. In contrast, the MIA1 offspring produced almost none of these behaviors.
Behavioral Observations
Autism is always diagnosed in humans by its behavioral characteristics. There is no objective or laboratory (e.g. blood) test for autism. The core symptoms of autism are
- repetitive, compulsive behaviors;
- reduced language and communicative ability; and
- reduced social interactions.
At ages of 10, 17, and 22 months, monkeys were subjected to tests for these autism behaviors.
Repetitive, Compulsive Behavior
Both first and second trimester immune activation caused large increases in repetitive/stereotyped behaviors. See A charts in figure below (top row).
Affiliative Vocalizations
“Affiliative” behaviors in monkeys are behaviors that reduce group tension and promote social bonding. Grooming is an example of an affiliative behavior. Affiliative vocalization is akin to friendly chatting among monkeys. Both first and second trimester immune activation caused significant decreases in affiliative vocalization. See B charts in figure below (middle row).
Social Interactions
Tested monkeys were observed while they interacted with other unfamiliar monkeys. The MIA1 monkeys displayed abnormal, inappropriate social interactions. Specifically, the MIA1 monkeys moved too close to unfamiliar monkeys, and remained awkwardly close to unfamiliar monkeys. See C charts in figure below (bottom row).
.
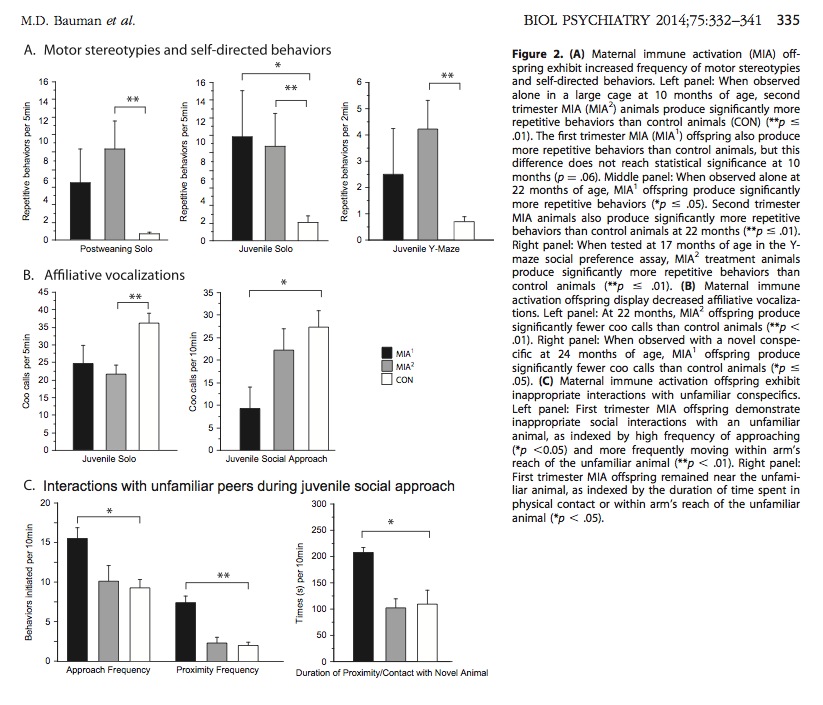
Above: Monkeys exposed to immune activation display repetitive behavior, reduced vocalization, and inappropriate social behavior. These are the three core symptoms of autism.
Increased pacing was also observed in MIA-exposed monkeys.
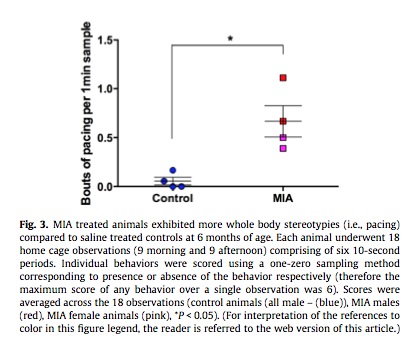
Above: Monkeys exposed to immune activation display greatly increased pacing behavior. Pacing is common in human autism.
Some of these behavioral changes increased as the monkeys matured. The paper states
Stereotypic behaviors, for example, are one of the diagnostic features of ASD and were consistently observed throughout postnatal development in the MIA2 offspring and to a lesser extent in the MIA1 offspring.
and
We suggest that the inappropriate social approach behaviors observed in the animal model may be reminiscent of the active but odd subtype of social interaction style described in autistic spectrum disorder and the complex social functioning impairments in schizophrenia.
ASD = Autism spectrum disorder.
Social Attention
Eye tracking was used to monitor how the monkeys looked at monkey faces. The duration of face-fixation, and location of the fixation (e.g. on the eye or nose) were measured.
The MIA1 monkeys displayed significantly shorter-duration fixation on monkey faces, and monkey eyes in particular.
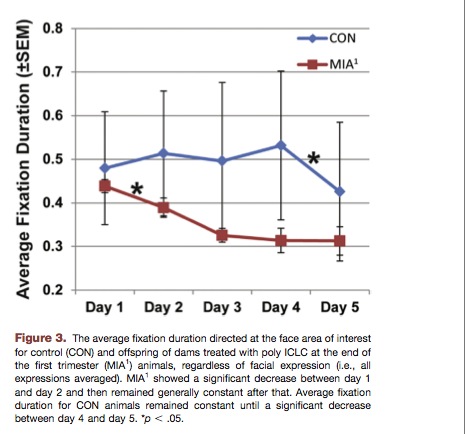
Above: Monkeys exposed to immune activation avoid looking at monkey faces. This is similar to human autism. Autistics tend to avoid looking at faces and eyes in particular.
Other changes in facial fixation were also observed. For example, MIA1 monkeys did not look at the eye when looking at monkey faces. See paper #2 for details. Paper #2 states
The lack of attention to the eye region exhibited by MIA1 animals represents a significant departure from social processing in normal macaques and typically developing humans and parallels findings in both autistic spectrum disorder and schizophrenia clinical populations.
and
Many individuals with autistic spectrum disorder also demonstrate abnormal fixation patterns when viewing faces, characterized by a lack of fixation in the eye region and a possible compensatory strategy of fixating on the mouth region.
Morphological Changes
Paper #3 provides the first preliminary results of investigation of the monkey brains. The monkeys were killed at ages 41-46 months.
Neuron morphology (physical features of dendrites) was analyzed. Immune activation was found to affect number of dendrites and dendrite diameter in specific locations on neurons.
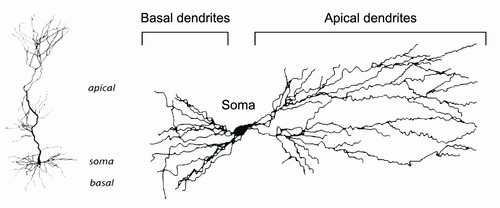
Above: Diagram of a neuron, showing the different types of dendrites. In the monkey experiments, immune activation affected some types of dendrites in specific places.
The authors state
In the limited 30 micron section of apical dendrite, at a distance of 100 ± 10 microns from the soma, the apical dendrites of MIA treated animals were smaller in diameter than those of the control animals (Z = 2.31, P < 0.05) (Fig. 4).
and
. . . we found a significantly larger number of oblique dendrites between the soma and the beginning of the selected section (Z = 2.14, P < 0.05) (Fig. 5).
Oblique dendrites = Dendrites that branch from apical dendrites. They are small “branches.”
It is unknown if these specific findings are present in human disease, but the authors state
The goal of the present study was to determine if this novel, nonhuman primate model of MIA model exhibits neuropathology relevant to human neuropsychiatric diseases. We elected to focus the initial evaluation of the nonhuman primate model in the DLPFC – a region of prefrontal cortex in humans and nonhuman primates that is essential for high order cognitive processes and implicated in social cognition (Arnsten, 2011). DLPFC dysfunction is strongly implicated in SZ (Barch and Ceaser, 2012; Lesh et al., 2013; Ursu et al., 2011) and to a lesser extent ASD (Morgan et al., 2012).
and
Although neuronal morphology in DLPFC has not been specifically evaluated in postmortem ASD brains, changes in pyramidal cell morphology have been described in other cortical regions.
DLPFC = dorsolateral prefrontal cortex, a region of the brain affected in schizophrenia, and possibly autism.
Pyramidal cell = neuron with triangular soma. Describes the shape of the cell.
SZ = schizophrenia.
ASD = autism spectrum disorder.
Here are the results discussed above:
Above: Immune activation caused changes in the dendrite diameter and number of oblique dendrites. These results may be relevant to schizophrenia and autism.
Conclusion
These findings clearly demonstrate that monkeys are affected by immune activation, just like mice. Monkeys exposed to immune activation display behaviors that closely resemble human autism and have physical signs of neuropathology.
It will be interesting to see if immune activation also causes the physiological damage observed in human autism and the mouse immune activation experiments: damaged Purkinje cells, mitochondrial dysfunction, dysbiosis and chronic brain inflammation.
Click here to go back to Part 2: Interleukin-6 and Autism.
Click here to see a listing of other blogs in this series.

















As a man, I certainly “get” the criticism of the name Thinking Moms’ Revolution. But I also see power and beauty in the name. Moms have gotten a bad rap from Freud onward. Imagine the trauma of taking your vaccine injured autistic child to the doctor and being told that you-a Refrigerator Mom-caused your child’s autism. (Has anyone heard of a Refrigerator Father being blamed for his child’s autism?)
Thinking Moms Revolution empowers moms to stand up to incompetent and far too often demeaning and abusive medical professionals.
Moms like the TMR moms model the strength and grit I want my three daughters to possess in a world that still tends to disempower women and mothers.
Thank you, Brett. We are definitely about empowering mothers to embrace the power they have to decide what happens to themselves and their children. xoxo
Sir , an honour to be hearing from you after that superb talk you gave in Florida (theTruthAboutHealth) - absolutely marvellous .
I might watch it again now because you have commented here .
London , England salutes you Sir .
TMR is wonderful .
The only criticism is why Mom is in the title . Why did that happen .
Should be renamed “Thinking Parents Revolution” .
Dads lives matter DLM
And there is a revolution coming , Brett Wilcox thinks so , and so do I .
😀 We agree that “Dads’ lives matter” too. And, yeah, I totally get your concern. The reason it started as “Moms” is because we mostly ran into other moms thinking about this stuff. We also feel that it is the traditionally “maternal” instinct to protect and nurture that is driving the revolution. We had a suggestion from a man to put a little “insert” into the logo that says “& Dads.” I like that idea, but we don’t have the money to update all the graphics and T-shirts.
… in the vast majority of cases, it’s the moms who are steering the autism recovery boat! It’s a common complaint. I wish more fathers would step up to the plate! Gold star to the dads out there who do take an active role in recovering their children from autism, and a super shiny gold star to the dads who are doing it alone!